Miniscule Particles with Massive Potentials
What's Hot in September/October 2011
by John Emsley
Graphene continues to dominate the Hot Ten, but there is a paper, #6, which manages to combine graphene chemistry with another popular topic: nanochemistry. It comes from a group at Pacific Northwest National Laboratory in Richland, Washington, led by Jun Liu, working in collaboration with Ilhan Aksay of Princeton University. Paper #6 concerns titanium dioxide-graphene hybrid nanocrystals, which could benefit lithium-ion storage batteries.
TiO2-graphene nanocomposites are not new, but the method of making them by Liu and colleagues is. It uses graphene sheets dispersed in an aqueous solution of the surfactant sodium dodecyl sulfate, and it is on these that nanocrystals of TiO2 are deposited. The process is a mild, low-temperature (below 100 °C) crystallization which results in either of the crystal forms of TiO2, namely rutile and anatase.
The nanocrystals varied slightly in size, with the rutile ones being 6 nm and the anatase ones 5 nm, as measured by transmission electron microscopy. Rutile crystals were in the majority under the conditions described above, although the distribution shifted in favor of anatase when sodium sulfate was added to the solution.
The effectiveness of these so-called functionalized graphene sheets (FGSs) was demonstrated as the electrodes of a typical Li-ion battery. Between 0.5 and 2.5% FGS was added to the electrodes, and this more than doubled the charge and discharge rates, the effect being attributed to the presence of the graphene network now embedded in the metal oxide electrodes.
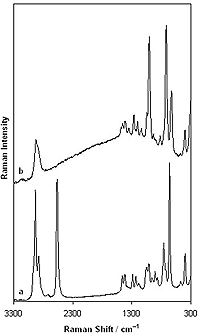
(a) below: Raman spectrum of liquid 2-mercaptoethanol
and (b) above: SERS spectrum of 2-mercaptoethanol monolayer formed on
roughened silver. SERS measurement was carried out for metal substrate
immersed in a 10 mM 2-mercaptoethanol aqueous solution. Spectra are scaled
and shifted for clarity. From the
Wiki Commons.
Paper #9 in the list is also about another remarkable effect due to nanoparticles: in this case, of gold nanoparticles coated with a layer of an oxide, either silica or alumina. These have a dramatic effect on the analytical technique known as surface-enhanced Raman spectroscopy (SERS)—so dramatic that it permits the characterization of individual molecules.
The Raman spectrum of a molecule arises from its vibrational modes of bonds within the molecule. SERS is a technique which has become widely used once it was realized that if the sample to be investigated was deposited on a nano-structured surface, such as gold or silver, the sensitivity would be enhanced a millionfold, even to the extent of permitting detection of a single molecule.
Now two groups of researchers, one based at Xiamen University, China, and headed by Zhong Qun Tian, the other at Georgia Institute of Technology, Atlanta, have collaborated and come up with SHINERS (a.k.a. shell-isolated nanoparticle-enhanced Raman spectroscopy). In this, the sample to be investigated is placed on the surface of a single crystal of platinum covered with a layer of hydrogen, and then dusted with a layer of the silica-coated gold nanoparticles. (The silica film acts to insulate the gold from direct contact with the sample.)
The result is a Raman spectrum of sufficient sensitivity to identify molecules of the pesticide parathion on an orange peel, as well as biomolecules on the surface of yeast cells.
And still on the theme of nanochemistry there is, just offstage at #11, a paper by Peidong Yang and Erik Garnett of the University of California, Berkeley, which could have significant implications for solar cells. (E. Garnett, P. Yang, Nano Lett., 10[3]: 1082-7, 2010; 32 citations this period, 63 overall.)
These two have demonstrated that ordered arrays of silicon nanowires have excellent light-trapping ability and that these can be fabricated easily and have efficiencies of between 5 and 6%. The method of fabrication consisted of dipping a silicon wafer into an aqueous suspension of silica beads, followed by deep reactive ion etching using each bead as an etch mask from which to grow the nanowires. The beads were then removed using hydrofluoric acid.
Finally, at #17, is another nano paper of an entirely different kind. This reports a nanoscale porous metal-organic framework (MOF) that could be used for drug delivery. The development comes from a 19-strong group based at various institutions in France and Korea, and led by Patricia Horcajada of the Institut Lavoisier, Versailles, France. (P. Horcajada, et al., Nature Mater., 9[2]: 172-8, 2010; 27 citations this period, 69 total.)
The MOFs were compounds of iron(III) carboxylates of acids such as muconic
acid and fumaric acid. Within their enormous cavities, various drugs such
as retroviral and anti-tumor compounds could be accommodated and delivered
more effectively to where they were needed.![]()
Dr. John Emsley is based at the Department of Chemistry, Cambridge University, U.K.
| What's Hot in Chemistry | |||
|---|---|---|---|
| Rank | Paper |
Cites This Period Mar-Apr 11 |
Rank Last Period Jan-Feb 11 |
| 1 | X.S. Li, et al., "Large-area synthesis of high-quality and uniform graphene films on copper foils," Science, 324(5932): 1312-4, 5 June 2009. [U. Texas, Austin; Texas Instruments, Dallas] *453TF | 125 | 3 |
| 2 | Y.Y. Liang, et al., "For the bright future—Bulk heterojunction polymer solar cells with power conversion efficiency of 7.4%," Adv. Materials, 22(20): E135-8, 25 May 2010. [U. Chicago, IL; Solarmer Energy Inc., El Monte, CA] *612IK | 99 | 1 |
| 3 | S. Bae, et al., "Roll-to-roll production of 30-inch graphene films for transparent electrodes," Nature Nanotech., 5(8): 574-8, August 2010. [8 South Korean, Singaporean, and Japanese institutions] *635BZ | 71 | 10 |
| 4 | Y.Y. Liang, et al., "Highly efficient solar cell polymers developed via fine-tuning of structural and electronic properties," J. Am. Chem. Soc., 131(22): 7792-9, 10 June 1009. [U. Chicago, IL; Solarmer Energy Inc., El Monte, CA] *460HD | 45 | 6 |
| 5 | B. Lim, et al., "Pd-Pt bimetallic nanodendrites with high activity for oxygen reduction," Science, 324(5932): 1302-5, 5 June 2009. [Washington U., St. Louis, MO; Brookhaven Natl. Lab., Upton, NY] *453TF | 43 | 8 |
| 6 | D.H. Wang, et al., "Self-assembled TiO2-graphene hybrid nanostructures for enhanced Li-Ion insertion," ACS Nano, 3(4): 907-14, April 2009. [Pacific Northwest Natl. Lab., Richland, WA; Princeton U., NJ] *439HR | 42 | † |
| 7 | C.H. Lu, et al., "A graphene platform for sensing biomolecules," Angew. Chem. Int. Ed., 48(26): 4785-5, 2009. [Fuzhou U., China; First Inst. Oceanography, Qingdao, China] *464GX | 37 | † |
| 8 | F.C. Krebs, T. Tromholt, M. Jorgensen, "Upscaling of polymer solar cell fabrication using full roll-to-roll processing," Nanoscale, 2(6): 873-86, June 2010. [Tech. U. Denmark, Roskilde] *608ML | 34 | 7 |
| 9 | J.F. Li, et al., "Shell-isolated nanoparticle-enhanced Raman spectroscopy," Nature, 464(7287): 392-5, 18 March 2010. [Xiamen U., China; Georgia Tech, Atlanta] *570FG | 34 | † |
| 10 | X.R. Wang, et al., "N-doping of graphene through electrothermal reactions with ammonia," Science, 324(5928): 768-71, 8 May 2009. [Stanford U., CA; U. Florida, Gainesville; Lawrence Livermore Natl. Lab., CA] *442HN | 33 | † |
| SOURCE: Thomson Reuters Hot Papers Database. Only papers indexed by Clarivate since May 2009 are tracked. A dagger indicates that the paper was not ranked in the Top Ten during the last period. In the event that two or more papers collected the same number of citations in the most recent bimonthly period, total citations to date determine the rankings. | |||
