Alzheimer’s Research Shifts Focus to Soluble Forms of Beta-Amyloid Protein
What's Hot in Medicine September/October 2010
By David W. Sharp
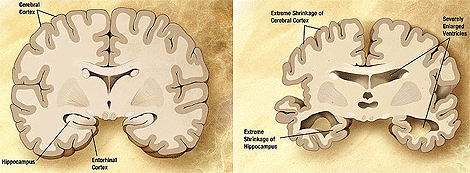
There are several forms of dementia. Alzheimer’s disease is just one of them. However, this is the most common type, and it is, thus far, an irreversible neurodegenerative condition with devastating consequences for patients and their families. Also, governments, faced with increasingly aging populations, are getting more and more anxious about the future provision of care for those affected. Drugs are being looked at, and some have been licensed, but the best that can be said for any of them is that they may slow down the rate of decline.
At a pathological level, a characteristic finding is the deposition in the brain of amyloid-beta protein, in the form of plaques and neurofibrillary tangles. The scale of those deposits appears not to be strongly associated with the amount of cognitive decline, and there has lately been a shift in emphasis from plaques and tangles to smaller, soluble forms of beta-amyloid.
One excellent review of soluble, non-fibrillar beta-amyloid assemblies has been provided by G.M.Shankar and D.M.Walsh (Mol. Neurodegener.,4: 48, 2009, and there is another by K. Broersen and colleagues (Alzheimers Res. Ther.,2[4], 2010: doi 10.1186/alzrt36).
Sometimes in citation rankings a paper appears low down, then drops out of the top league only to reappear higher up later. The article by Dr. Ganesh M. Shankar and colleagues (#8) has had that pattern; we first saw it in mid-2009, when it was at #21. This study was done on the rat hippocampus but made use of soluble beta-amyloid oligomers from the cerebral cortex of patients with Alzheimer’s disease. Relevant effects on the rat hippocampus were seen with soluble beta-amyloid but not with insoluble beta-amyloid plaque unless this was first solubilized.

Carol Brayne from University of Cambridge, U.K.
The soluble human beta-amyloid also interfered with the memory of rats’ learned behavior. Co-administration of antibody to the N-terminus of beta-amyloid prevented effects on the hippocampus. The authors see their findings as showing that beta-amyloid from human brains is "the most pathophysiologically relevant material ... for preclinical validation of agents designed to neutralize [beta-amyloid] aggregates."
If there is to be progress in the treatment of patients with Alzheimer’s disease it will come, I believe, from collaborative and multidisciplinary efforts. To try and illustrate the point, I looked at very recent papers coauthored by Carol Brayne, though no doubt many other researchers’ names would have served my purpose. She is professor of public health medicine at the University of Cambridge, U.K., and a study of the impact of recent guidelines on how drugs licensed for the treatment of Alzheimer’s disease should be used within the U.K. National Health Service clearly fits into a public-health focus (F.E. Matthews, et al., Int. J. Geriatr. Psychiatry,25[7]: 719-24, 2010).
A completely different paper of hers, also from this year, looks at the question intimated at the start of this column. If insoluble beta-amyloid does not correlate well with dementia, do soluble forms do any better? A study in 19 individuals without dementia and 24 with (Alzheimer pathology in 14 but not in the other 10) confirms that they do (J.M. Mc Donald, et al., Brain,133: 1328-41, 2010).
Professor Brayne does not feel qualified to explain the citation ups and
downs attached to #8 but she notes the growing interest in soluble
beta-amyloid as an avenue for research. "A good animal model result will
clearly be quoted when that avenue begins to look more promising in
humans," she tells Science Watch, adding, "We are at that point
now." Also, there is concern about how well animal models of degenerative
disorders reflect the human disorder. The toxicity of protein aggregates
(as in #8 and other papers), she notes, "is a much neater model than
looking for replications of complex neuropathological and neurobehavioral
profiles."![]()
A former deputy editor of The Lancet, David W. Sharp, M.A. (Cambridge) is a freelance writer living in Minchinhampton, U.K.
| What's Hot in Medicine | |||
|---|---|---|---|
| Rank | Paper |
Cites This Period Mar-Apr 10 |
Rank Last Period Jan-Feb 10 |
| 1 | The ACCORD Study Group (H.C. Gerstein, et al.), "Effects of intensive glucose lowering in type 2 diabetes," New Engl. J. Med., 358(24): 2545-59, 12 June 2008. [Writing Group: 10 U.S. and Canadian institutions] *311IJ | 89 | 2 |
| 2 | The ADVANCE Collaborative Group (A. Patel, et al.), "Intensive blood glucose control and vascular outcomes in patients with type 2 diabetes," New Engl. J. Med., 358(24): 2560-72, 12 June 2008. [Writing Group: 18 institutions worldwide] *311IJ | 87 | 3 |
| 3 | Novel Swine-Origin Influenza A (H1N1) Virus Investigation Team (F.S. Dawood, et al.), "Emergence of a novel swine-origin influenza A (H1N1) virus in humans," New Engl. J. Med., 360(25): 2605-15, 18 June 2009. [Writing group: Ctrs. for Disease Control & Prevent., Atlanta, GA] *458WR | 80 | 1 |
| 4 | R.R. Holman, et al., "10-year follow-up of intensive glucose control in type 2 diabetes," New Engl. J. Med., 359(15): 1577-89, 9 October 2008. [6 U.K. institutions] *358FS | 67 | 7 |
| 5 | C.L. Ogden, M.D. Carroll, K.M. Flegal, "High body mass index for age among US children and adolescents, 2003-2006," JAMA, 299(30): 2401-5, 28 May 2008. [Ctrs. for Disease Control & Prevent., Atlanta, GA] *305BD | 62 | 8 |
| 6 | NICE-SUGAR Study Investigators (S. Finfer, et al.), "Intensive versus conventional glucose control in critically ill patients," New Engl. J. Med., 360(14): 1283-97, 26 March 2009. [Writing Committee: 4 Australian, New Zealand, and Canadian institutions] *423VP | 54 | 9 |
| 7 | J.M. Llovet, et al., "Sorafenib in advanced hepatocellular carcinoma," New Engl. J. Med., 359(4): 378-90, 24 July 2008. [22 institutions worldwide] *329FK | 53 | 5 |
| 8 | G.M. Shankar, et al., "Amyloid-ß protein dimers isolated directly from Alzheimer’s brains impair synaptic plasticity and memory," Nature Medicine, 14(8): 837-42, August 2008. [5 U.S. and Irish institutions] *334QY 53 + | 53 | † |
| 9 | Cancer Genome Atlas Research Network (L. Chin, et al.), "Comprehensive genomic characterization defines human glioblastoma genes and core pathways," Nature, 455(7216): 1061-8, 23 October 2008. [60 institutions worldwide] *363FG | 50 | 10 |
| 10 | F.H. Schröder, et al., "Screening and prostate-cancer mortality in a randomized European study," New Engl. J. Med., 360(13): 1320-8, 26 March 2009. [15 institutions worldwide] *423VP | 49 | 4 |
|
SOURCE: Thomson Reuters Hot Papers Database. Read the Legend. |
|||
