Jaheon Kim on Highly Porous Metal-Organic Frameworks
Fast Breaking Papers Commentary, August 2011

|
Article: Ultrahigh Porosity in Metal-Organic Frameworks
Authors: Furukawa, H;Ko, N;Go, YB;Aratani, N;Choi, SB;Choi,
E;Yazaydin, AO;Snurr, RQ;O'Keeffe, M;Kim,
J;Yaghi, OM |
Jaheon Kim talks with ScienceWatch.com and answers a few questions about this month's Fast Breaking Paper paper in the field of Chemistry.
![]() Why do you think your paper is highly
cited?
Why do you think your paper is highly
cited?
The paper describes how to design and actually realize extremely porous metal-organic frameworks (MOFs). Research interest in crystalline porous solids has increased enormously in the last 15 years because these materials can be used in various applications such as catalysis, sorbents, separation, sensors, drug delivery, fuel cells, and hydrogen storage. These applications directly rely on the MOFs' porosity, and the paper touches this challenge. One of the MOFs in the paper achieved the world-record surface area of 6240 m2/g.
![]() Does it describe a new discovery, methodology, or
synthesis of knowledge?
Does it describe a new discovery, methodology, or
synthesis of knowledge?
The paper describes and demonstrates the production of MOFs having a huge empty space with very thin walls. Nonetheless, they are robust enough to endure a vacuum when their pores are fully evacuated. MOFs are built from two basic components: metal clusters and organic linkers. Various connections of these simple building blocks have produced over 3,000 MOFs so far.
To enlarge their storage space and surface areas, MOFs' struts must be fully accessible to guests, which can be achieved through making the frameworks as thin and light as possible. The frameworks should also avoid interpenetration, which is the establishment of frameworks inside the spaces provided by the initial framework—a frequently observed phenomenon in MOFs.
|
|
|
|
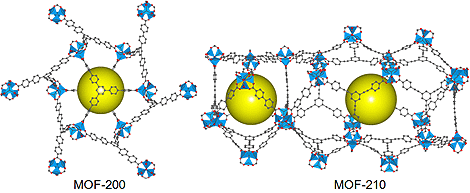
FIGURE DESCRIPTION: Crystal structures of MOF-200 and MOF-210. Prepared by Jaheon Kim. View two additional figures and descriptions of Jaheon Kim's work. |
|
|
|
We demonstrated two widely applicable strategies: one is the isoreticular expansion of a known framework (MOF-200) which never forms an interpenetrated structure. The other is producing a new network using two types of organic linkers where the ratio of the linker length is the key for the success (MOF-210).
![]() Would you summarize the significance of your paper
in layman's terms?
Would you summarize the significance of your paper
in layman's terms?
To date, although increasing attention has been paid to hydrogen and methane as alternative fuels of automobiles, there is a challenge to realize a gas storage system which can keep gases effectively and safely. We have discovered and noticed that the metal-organic framework (MOF) is one of the excellent crystalline porous materials to meet such requirements. This is because, in the MOF structure, metal clusters are connected by organic linkers to form infinitely expanded 3D structure so that it is possible to provide the large accessible surface without losing the structural integrity.
In this study we showed that well-tuned MOFs whose pore diameters are 2-3 times larger than the diameter of gas molecules are the optimal materials to maximize the gas storage capacity. Indeed, the surface area in one gram of MOF is even larger than the area of football field. Thanks to the ultrahigh surface area, these MOFs can store 2-3 times large amounts of hydrogen, methane, and nitrogen as well as carbon dioxide. We believe that these materials pave the way for the storage and transportation of clean energy.
Porous solids are useful in many applications such as catalysis, separation, sensing, and storage. Their pore sizes range from a few angstroms up to micrometers depending on their constituent components. Modulation of pore size and functionality at a molecular level can allow full exploitation of these materials for use in various applications. Regardless of their constituent components, porous materials such as zeolites, carbons, MOFs, etc., share a common physical property in that their surface area represents how much the inner spaces of the bulk particles can be used as pores.
By designing and synthesizing MOFs, the paper describes how to maximize this key property of crystalline porous solids. For example, MOF-210 has the surface area of 6240 m2/g—i.e., one gram of the material has inner surfaces covering over 78 m × 78 m. MOF-200 has the same pore volume of 3.6 cm3/g as that of MOF-210. The high surface areas and large pore volumes allow these materials to store large amounts of methane, carbon dioxide, or hydrogen gases when filled in cylinders.
![]() How did you become involved in this research, and
how would you describe the particular challenges, setbacks, and
successes that you've encountered along the way?
How did you become involved in this research, and
how would you describe the particular challenges, setbacks, and
successes that you've encountered along the way?
"Our materials can be used for storing fuel gases or capturing greenhouse gases, contributing to the development of a clean energy society."
Research in this field over the last decade has been characterized by the quest for ultrahigh porous MOFs, with higher surface areas ever being sought. Omar M. Yaghi's group at UCLA and my group have challenged this problem. Based on experience in synthesizing designer MOFs, we tried to make very porous MOFs through two conditions: 1) using thin and long organic building block frameworks, and 2) adopting non-interpenetrating nets. This allowed us to make the ultrahigh porous MOFs described in the paper.
To verify the procedures and their outcomes, we had to overcome several obstacles. The first was to elucidate the X-ray structures of crystals that essentially comprise empty space. The second was to evacuate the MOFs properly to demonstrate the presence of permanent porosity. Dr. Furukawa in the UCLA group assessed the ultrahigh porosity of MOF-200 and MOF-210 using gas adsorption measurements, which was also supported by theoretical calculations conducted by the Snurr group at Northwestern University. The success was achieved through consistent collaboration among research groups.
![]() Where do you see your research leading in the
future?
Where do you see your research leading in the
future?
In the near future, many other ultrahigh porous MOFs will be explored because it was shown that the ultrahigh porosity could be achieved by two very different MOF structures. Future work will aim to introduce specific functionality to the regular pores of crystalline materials; i.e., various organic functional groups will be introduced into MOF platforms without disturbing the original framework connectivity.
Anticipated applications include encapsulating functional nanoparticles or quantum dots for catalysts, sensors, or light-emitting materials, storing fuel gas such as methane and hydrogen—in particular, the cryogenic storage of hydrogen, storing large functional biomolecules such as enzymes, and anchoring synthetic catalysts for various chemical reactions.
![]() Do you foresee any social or political
implications for your research?
Do you foresee any social or political
implications for your research?
Our materials can be used for storing fuel gases or capturing greenhouse
gases, contributing to the development of a clean energy society. Like
medicines for human health, these energy materials can help sustain our
environment's health. There are no political implications.![]()
Jaheon Kim, Ph.D.
Department of Chemistry
Soongsil University
Seoul, South Korea
KEYWORDS: METAL-ORAGANIC FRAMEWORKS, ULTRAHIGH POROSITY, HYDROGEN STORAGE, SURFACE AREAS, COORDINATION POLYMERS, ADSORPTION, DESIGN, ROUTE, SITES.
