In Prognosis and Treatment, Genome Studies Continue to Enlighten
What's Hot in September/October 2011
by David W. Sharp
In just seven weeks between the beginning of June 2011 and mid-July appeared at least 20 research papers on a single-nucleotide polymorphism (SNP) lying on chromosome 19 close to the gene (IL28B) that encodes a type III interferon. This is an astonishing rate of publication, a trigger for which must surely have been the contribution now standing at #6 in the Top Ten, with an accumulated 279 citations in the Hot Papers database as of March-April 2011.
This publication showed that genetic variation in IL28B can help clinicians and patients predict response to treatment (pegylated interferon and ribavirin being the current standard) given for hepatitis C. A companion paper now stands at #25 (D.L. Thomas, et al., Nature, 461[7265]: 798-801, 2009; total cites 160, latest count 43), this being concerned with the natural history of hepatitis C, whereas the report from three weeks earlier in 2009 (#6) looked at sustained viral clearance in response to treatment.
The SNP, known as rs12979860, was identified via a genome-wide association study. Genetic markers like this need confirmation, and for rs12979860 and hepatitis C this came rapidly and consistently. For example, a statistical exercise revealed genotype CC to be the strongest predictor of response to antiviral treatment (T.R. O’Brien, et al., PLoS One, 6[7]: e20904, 2011 [Epub July 8, 2011]). The genotyping can now be done rapidly (e.g., R. Melis R, et al., J. Mol. Diag., 13[4]: 446-51, 2011), and within a year of the seminal publication the case was being made for IL28B genotyping being built into the design for trials of hepatitis C treatments.
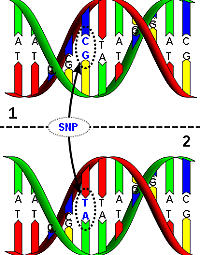
DNA molecule 1 differs from DNA molecule 2 at a single
base-pair location (a C/T polymorphism),
Wiki Commons.
The previous Top Ten commentary in July/August 2011touched on a new drug for melanoma (vemurafenib or PLX4032) which had attracted attention at an oncology meeting earlier this year. The topic deserves more space because at #11 this time we have a phase I study of this drug (K.T. Flaherty, et al., New Engl. J. Med.,363[9]: 809-19, 2010; total cites 116, latest count 62).
Roughly half of all melanomas carry a mutation in the gene that codes for a kinase called BRAF, and vemurafenib is a powerful inhibitor of BRAF with this mutation. At least three other drugs with the same target are being looked at. Although the study focused on drug dosage and toxicity, encouraging tumor responses were recorded. A phase III clinical trial was reported to be in progress—indeed, we now have the six-month results from that (P.B. Chapman, et al., New Engl. J. Med., 364[26]: 2507-16, 2011).
Six hundred and seventy-five patients with previously untreated metastatic melanoma and possessing the specific mutation took part in this randomized comparison of vemurafenib and dacarbazine. Overall survival was 84% for patients on the new drug and 64% for the control group, and the trial monitoring committee advised switching those on dacarbazine to vemurafenib. Drug approval is being sought from U.S. and European licensing agencies, and a rapid diagnostic test for the mutation has been developed.
If vemurafenib does prove to be the breakthrough that we all hope for, this will provide huge encouragement to everyone involved in the development of genetically tailored cancer treatments, where there have been so many ups and downs. Vemurafenib itself is no exception to this "roller coaster of breakthroughs and setbacks" (as recorded in Amy Harmon’s New York Times series on this drug in February, 2010).
This development has involved basic-research companies and the
pharmaceutical industry working together—indeed, Roche (veramufenib)
and Bristol-Myers Squibb (ipilimumab, the anti-melanoma agent discussed in
ScienceWatch.com
last month) are collaborating on a two-agent approach. While stock
exchanges quiver with excitement, let us not forget where the story really
began and when—just nine years ago with a publication from the
U.K.-based Cancer Genome Project funded by a medical research charity, the
Wellcome Trust (H. Davies, et al., Nature, 41[6892]:
949-54, 2002).![]()
A former deputy editor of The Lancet, David W. Sharp, M.A. (Cambridge), is a freelance writer living in Minchinhampton, Gloucestershire, U.K.
| What's Hot in Medicine | |||
|---|---|---|---|
| Rank | Paper |
Cites This Period Mar-Apr 11 |
Rank Last Period Jan-Feb 11 |
| 1 | Novel Swine-Origin Influenza A (H1N1) Virus Investigation Team (F.S. Dawood, et al.), "Emergence of a novel swine-origin influenza A (H1N1) virus in humans," New Engl. J. Med., 360(25): 2605-15, 18 June 2009. [Writing group: Ctrs. for Disease Control & Prevent., Atlanta, GA] *458WR | 121 | 1 |
| 2 | K.M. Flegal, et al., "Prevalence and trends in obesity among US adults,1999-2008," JAMA, 303(3): 235-41, 20 January 2010. [Ctrs. Disease Control & Prevent., Hyattsville, MD] *544VM | 109 | 6 |
| 3 | T.S. Mok, et al., "Gefitinib or carboplatin-paclitaxel in pulmonary adenocarcinoma," New Engl. J. Med., 361(10): 947-57, 3 September 2009. [14 institutions worldwide] *490EI | 106 | 5 |
| 4 | R.J. Garten, et al., "Antigenic and genetic characteristics of swine-origin 2009 A(H1N1) influenza viruses circulating in humans," Science, 325(5937): 197-201, 10 July 2009. [26 institutions worldwide] *468FK | 88 | 3 |
| 5 | R. Perez-Padilla, et al., "Pneumonia and respiratory failure from swine-origin influenza A (H1N1) in Mexico," New Engl. J. Med., 361(7): 680-9, 13 August 2009. [Natl. Inst. Respiratory Diseases, Mexico City; BIRMEX, Mexico City; Secretariat of Health, Mexico City] *482JX | 82 | † |
| 6 | D.L. Ge, et al., "Genetic variation in IL28B predicts hepatitis C treatment-induced viral clearance," Nature, 461(7262): 399-401, 17 September 2009. [Duke U., Durham, NC; Schering-Plough Res. Inst., Kenilworth, NJ; Johns Hopkins U., Baltimore, MD] *494QB | 82 | † |
| 7 | S.J. Connolly, et al., "Dabigatran versus warfarin in patients with atrial fibrillation," New Engl. J. Med., 361(2): 1139-51, 17 September 2009. [12 institutions worldwide] *494QA | 76 | 4 |
| 8 | S. Jain, et al., "Hospitalized patients with 2009 H1N1 influenza in the United States, April-June 2009," New Engl. J. Med., 361(20): 1935-44, 12 November 2009. [Ctrs. for Disease Control & Prevent., Atlanta, GA] *518AI | 72 | 2 |
| 9 | P.C. Fong, et al., "Inhibition of poly(ADP-ribose) polymerase in tumors from BRCA mutation carriers," New Engl. J. Med., 361(2): 123-34, 9 July 2009. [7 U.K.. and European institutions] *467OS | 71 | † |
| 10 | K. Hancock, et al., "Cross-reactive antibody responses to the 2009 pandemic H1N1 influenza virus," New Engl. J. Med., 361(20): 1945-52, 12 November 2009. [Ctrs. for Disease Control & Prevent., Atlanta, GA] *518AI | 65 | 9 |
| SOURCE: Thomson Reuters Hot Papers Database. Only papers indexed by Clarivate since May 2009 are tracked. A dagger indicates that the paper was not ranked in the Top Ten during the last period. In the event that two or more papers collected the same number of citations in the most recent bimonthly period, total citations to date determine the rankings. | |||
